Dislocations move through a crystal lattice much like a ridge can be moved across a carpet, slowly moving the carpet across a floor without moving the whole carpet at one time. Their motion accounts for much of the strain in crystals. The presence of dislocations weakens the crystal structure. They are often referred to as an extra half-plane because of this geometry. Different slip systems may be activated at different temperatures, or strain rates, so the relationships of which slip systems are active under different deformation conditions are complex.ĭislocations are best thought of as an extra plane of atoms that terminates somewhere in the crystal lattice. If the crystal has fewer than five, it will eventually crack or fracture. This increase in stress is known as work hardening.Ĭrystals need to have five independent slip systems to accommodate any general homogenous strain. soon different slip systems start to interfere with each other and deformation will either stop or the stress will rise to enable continued deformation. Most crystals have many slip systems activated at different critical resolved shear stresses, so slip begins on the planes with the lowest critical resolved shear stress. Slip on these crystallographic directions begins when the critical resolved shear stress for that crystallo-graphic direction is surpassed. occur in directions that do not juxtapose ions of like charge.have short distances between equivalent atoms.slip, or translation gliding, usually occurs in very specific crystallographic directions in crystals called slip systems, favoring directions that Translation gliding is a deformation mechanism in crystals whereby the crystal lattice structure slips along some internal crystallographic plane, after some critical value of shear stress is reached. The amount of strain is limited by the crystallographic relationship for each type of crystal, though it typically falls in the range of 20°-45°. The shear occurs across a crystal plane, which must be a symmetry plane of the crystal. Mechanical or stress-induced twinning differs from growth twinning in that shear across a crystal plane changes the lattice orientation. 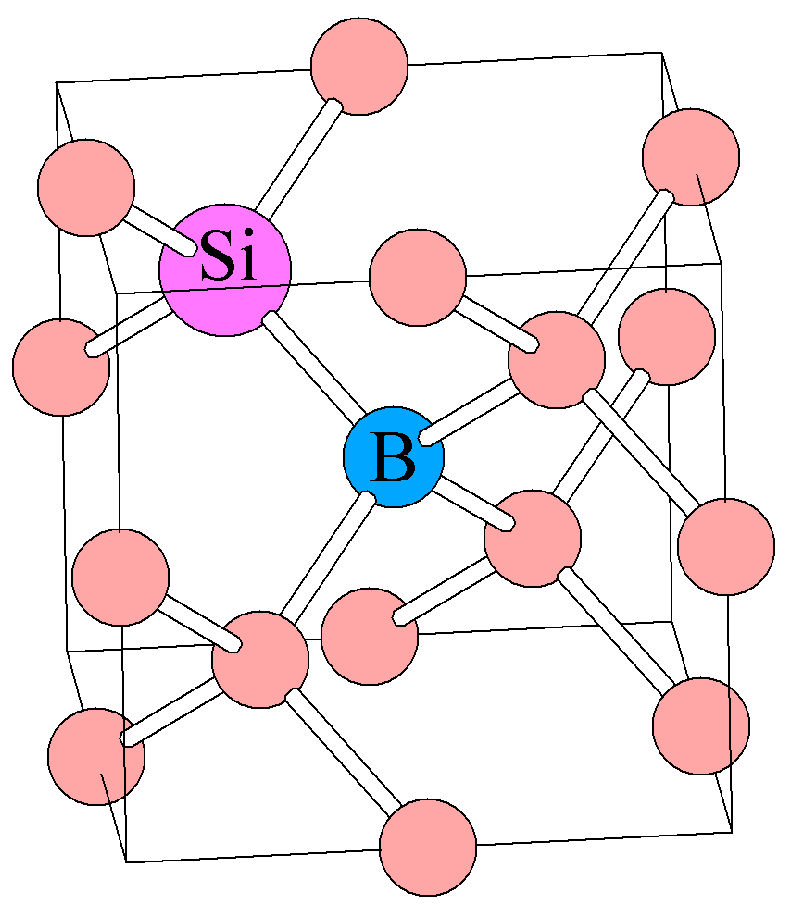 
Twinning, the misorientation of a crystallo-graphic plane across a plane in the crystal lattice, can be caused by mismatched growth or by deformation. This kind of diffusion can accommodate a general shape change of a crystal, such as occurs during regional deformation in many mountain belts worldwide. This process of migration of vacancies is called Nabarro-Herring creep. When a crystal is stressed, vacancies tend to move, or diffuse, in an orderly manner related to the stress field, migrating toward the crystal face with the highest stress, whereas atoms tend to migrate in the opposite direction toward the crystal faces with the lowest stress. Temperature causes the number of vacancies to increase, whereas pressure causes the number of vacancies to decrease. To react or deform when they have a higher internal energy. Crystals are more aptĬubic crystal of fluorite, about two inches (5 cm) on each face, on quartz with barite from Frazer's Hush Mine, Weardale, England (MarkA. Crystals having point or other defects have more internal energy because many bonds are broken or unsatisfied and the electric charges are not neutralized. In a regularly ordered crystal lattice most electric charges are satisfied by bonding and balancing positive and negative charges. Other types of point defects are more complex and involve more than one atom at a time. several types include impurities, in which the wrong type of atom is present in the crystal lattice in the place of another vacancies, in which an atom is missing from the atomic lattice and interstitials, in which an atom occupies a site that is not normally occupied. Point defects are considered irregularities, or defects, that affect one point in a crystal lattice. Point defects include vacancies, impurities, and interstitials, whereas line defects are known as dislocations. There are two main types of crystal defects: point defects and line defects. The motion of crystal defects is an important deformation mechanism, with different types of motion of different types of defects operating at different temperatures, pressures, and applied stresses. The motion of these defects accommodates the strain of crystals. 
These defects determine the strength of crystals, minerals, and rocks. Some defects are acquired during growth of the crystals when they first form, and others develop in the crystals during deformation. Crystals are regularly ordered symmetrical arrays of atoms, but like anything else in nature they are not perfect and have many defects. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
